Cancer often feels like a mysterious enemy that grows out of nowhere and keeps coming back no matter what treatments doctors throw at it. At the heart of this puzzle lies something called clonal populations, a concept that explains how most cancers begin, grow, and evolve. In everyday language, a clonal population is simply a large group of cells that all trace their family tree back to one single original cell. These cells are genetically very similar because they are direct descendants, like cousins in a huge extended family who inherited the same quirky traits from a shared grandparent. When this process goes wrong in the body, it turns into the engine that drives cancer. Understanding clonal populations in cancer helps explain everything from why tumors form to why standard treatments sometimes fail and why researchers are now designing smarter therapies. This guide breaks it down without complicated science, addressing the most common questions that arise when people encounter the term after a diagnosis or pathology report, particularly in relation to frequently discussed cancers such as breast cancer, lung cancer, leukemia, colorectal cancer, and others where clonal dynamics play a central role.
If any part of this explanation leaves you wanting more clarity, especially about your own pathology report, services like Honest Pathology offer virtual consultations with board-certified pathologists who review reports line by line, explain terms in plain language, and answer your specific questions directly.
What Is a Clonal Population in Cancer?
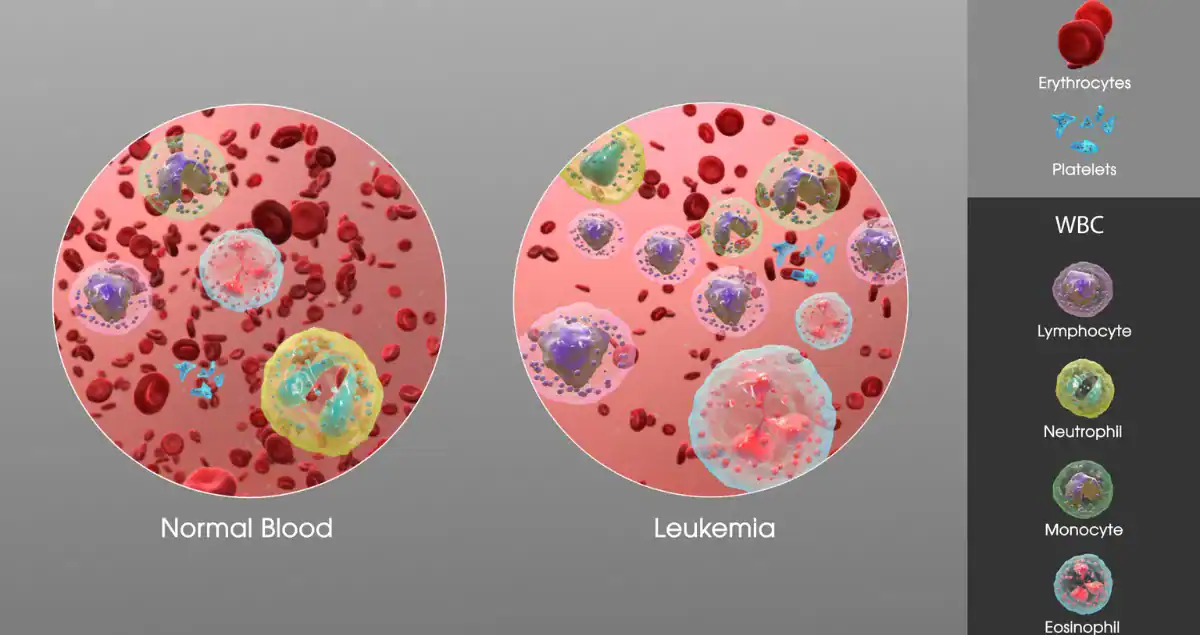
Think of your body as a city full of normal cells going about their daily business. Every cell divides now and then to replace old ones, but each new cell is supposed to stay identical to its parent only in the ways that keep everything running smoothly. A clonal population starts when one single cell picks up a damaging mutation in its DNA. That mutation gives the cell an unfair advantage, such as the ability to divide faster or ignore signals telling it to die. Instead of dying off like a normal cell would, this rogue cell begins copying itself over and over. All the copies inherit the exact same mutation, creating a growing crowd of identical troublemakers. In cancer, nearly every tumor begins this way. Scientists call the original troublemaker the founding clone because it launches the entire process. The group that descends from it is the clonal population. This is why pathologists often describe cancers as monoclonal, meaning they come from one clone rather than many unrelated cells starting the problem at once. For the vast majority of cancers the answer is yes, every cancer is clonal at its core. Even when tumors appear messy and varied under a microscope, they almost always share those core mutations passed down from that first mutated cell.
How Do Clonal Populations Start and Expand in the Body?
Clonal expansion is the next step, and it is exactly what it sounds like: the clone gets bigger and bigger until it crowds out healthy tissue. Picture a single weed seed landing in a perfect lawn. At first it is tiny and harmless. But if the weed has a mutation that lets it grow faster or resist the lawnmower, it spreads, sending out runners and seeds that create more identical weeds. Soon the lawn is patchy and the weed population dominates. In the body, the same thing happens with cells. Normal cells have built-in brakes that stop them from dividing too much or invading other areas. A mutation knocks out one of those brakes, so the cell and all its descendants keep multiplying. Over time this clonal expansion creates a lump, or tumor. The process can take years. In blood cancers such as leukemia, the expanding clone can slowly replace normal blood cells, leading to fatigue, infections, or bleeding problems. The first mutation can arise from random errors during cell division, exposure to chemicals or radiation, inherited risks, or simply aging. The key point is that once the clone starts expanding, the body’s normal checks and balances no longer apply to these cells, which is why clonal populations in cancer often feel unstoppable at first glance.
The Process of Clonal Evolution in Cancer Tumors
Clonal populations do not stay the same forever. This is where clonal evolution enters the picture, and it explains why cancer can change and become harder to treat over time. Just as animals evolve by natural selection, cancer cells keep mutating and the fittest ones survive. The original clone keeps dividing, but every division carries a small chance of new mistakes in the DNA. Most extra mutations are harmless or even harmful to the cell, so those cells die off. Occasionally, however, a new mutation gives one cell an extra advantage—such as the ability to hide from the immune system or survive chemotherapy. That cell and its descendants form a new subclone that begins to outgrow the others. Over months or years the tumor becomes a mix of related but slightly different clonal populations, each competing for space, nutrients, and survival. This evolutionary process explains why a cancer that responds well to initial treatment can return in a more aggressive form. The treatments kill off the weaker clones, but any rare resistant subclones that were already present suddenly face no competition and expand rapidly. Clonal evolution in cancer follows the same survival-of-the-fittest logic that Darwin described for species, only happening inside one person’s body at lightning speed.
Linear and Branched Clonal Evolution Explained
Cancer researchers describe two main patterns of how clonal populations evolve. In linear evolution the changes happen in a straight line, like steps on a ladder. Each new helpful mutation takes over completely, so the tumor at any moment looks fairly uniform because one clone replaces the previous one. This pattern is simpler but less common in advanced cancers. Branched evolution is more like a tree with many limbs growing at the same time. Different cells in the original clone pick up different extra mutations, and several subclones grow side by side. The tumor ends up with a lot of genetic diversity, which makes it much harder to wipe out. One branch might be sensitive to a drug while another branch is completely resistant. Both patterns start from the same founding clonal population, but the branched version creates the messy, heterogeneous tumors that doctors see in scans and biopsies. Branched patterns appear frequently in cancers such as chronic lymphocytic leukemia, multiple myeloma, and many solid tumors, and they often correlate with faster progression and poorer outcomes because the tumor has more options for survival.
Clonal Populations Versus Subclones: Understanding Tumor Diversity
A common question after reading a pathology report concerns the difference between clonal and subclonal. The main clonal population carries the original mutations that every cancer cell in the tumor shares; these are the trunk mutations present from the very beginning. Subclones are the smaller branches that carry extra private mutations on top of the trunk ones. In a biopsy, doctors can now measure how much of the tumor is the dominant clone versus the various subclones. High diversity, meaning many different subclones, usually signals a more dangerous cancer because it has more ways to adapt. This intratumor heterogeneity is one reason two patients with the same type of cancer can respond so differently to the same treatment. One patient’s tumor might be mostly one clone while another’s has dozens of competing subclones. Understanding this distinction helps explain why targeted drugs that hit a trunk mutation sometimes work beautifully at first but later fail when subclones that never had that mutation take over. For personalized insight into what your report shows about clonal diversity, resources like Honest Pathology provide expert reviews that break down these details clearly.
Clonal Evolution in Specific Cancers
Clonal dynamics are especially well documented in several common cancers. In breast cancer, particularly triple-negative or advanced cases, tumors often show branched clonal evolution with high intratumor heterogeneity, which contributes to resistance after initial therapy and explains why recurrences can behave differently from the original tumor. Lung cancer, especially non-small cell types such as adenocarcinoma, frequently displays extensive clonal diversity; subclones can drive metastasis or resistance to targeted drugs like EGFR inhibitors, with pre-existing resistant clones expanding under treatment pressure. Leukemia, including acute myeloid leukemia (AML) and chronic lymphocytic leukemia (CLL), is a classic example—clonal evolution here is often linear or branched, with subclones acquiring poor-prognosis mutations like those in TP53, making relapse common after chemotherapy. Colorectal cancer shows striking clonal evolution during metastasis; the founding clone from the primary tumor survives to spread, but new subclones emerge in distant sites or under therapy like anti-EGFR drugs, leading to rapid resistance via mutations in RAS or BRAF pathways. Other examples include prostate cancer and head and neck cancers, where branched heterogeneity correlates with poorer survival, and multiple myeloma, where neutral or branched evolution links to aggressive disease. These patterns highlight why clonal analysis is increasingly used to guide therapy in these cancers.
Why Clonal Populations Make Cancer Difficult to Cure
The biggest frustration for patients and doctors is treatment resistance, and clonal populations are the reason. Chemotherapy or targeted therapy acts like a weed killer that wipes out the easy-to-kill clones. Any surviving subclones that already carried resistance mutations suddenly face no competition and expand into a new dominant clonal population. This is clonal evolution in action under treatment pressure. It also explains why metastatic cancer is so hard to control: when cells spread to new organs, different environments favor different subclones, so the cancer at each site can evolve independently. The answer to why cancer often returns after remission almost always traces back to tiny undetected subclones that were present from the start. The good news is that knowing this is driving new strategies. Doctors now try to use combination therapies that attack multiple clones at once, or drugs that keep all clones under control without giving any one the chance to take over.
Clonal Hematopoiesis: When Clonal Populations Appear in Healthy Blood
Not every clonal population immediately means cancer. A closely related condition called clonal hematopoiesis, often shortened to CHIP or CH, is something many older adults have without knowing it. In this case, blood stem cells in the bone marrow acquire mutations and expand into a clonal population of blood cells. It is not cancer yet, but it raises the risk of developing blood cancers like leukemia later in life. Clonal hematopoiesis becomes more common with age because the older the stem cells get, the more likely they are to pick up DNA mistakes. Doctors find it incidentally during genetic testing done for other reasons. The risk is real but still low for most individuals; only a small percentage ever progress to actual cancer. However, it also links to higher chances of heart disease and poorer outcomes if cancer develops elsewhere in the body, including solid tumors like lung, breast, prostate, or colon cancer. Monitoring clonal hematopoiesis helps doctors catch problems early.
How Doctors Detect and Study Clonal Populations Today
Detecting clonal populations used to rely on simple chromosome tests or looking at cell shapes under a microscope. Today, advanced DNA sequencing can read the mutations in thousands of individual cells, mapping out the entire family tree of clones and subclones. This technology shows doctors exactly which mutations sit on the trunk versus the branches and how big each clonal population is inside the tumor. Newer single-cell techniques even reveal what each clone is doing functionally, not just what mutations it carries. These tools are turning clonal evolution from a scientific curiosity into a practical roadmap for treatment. Patients now receive answers based on real clonal architecture rather than guesswork.
What This Means for Cancer Treatment and Prevention
Knowing that cancer is driven by clonal populations changes everything about how we fight it. Instead of aiming only to shrink the visible tumor, researchers now design therapies that target the founding clone mutations present in every cell or combinations that hit multiple subclones simultaneously. Some approaches try to prevent clonal expansion in the first place by catching precancerous clonal populations early, especially in clonal hematopoiesis. Lifestyle factors that reduce DNA damage, such as avoiding smoking and limiting alcohol, indirectly slow the chance of new clones forming. For those already diagnosed, understanding clonal populations empowers better questions at the oncology office: Is my tumor branched or linear? Are there detectable resistant subclones? What treatment attacks the trunk mutations? The future of cancer care lies in staying one step ahead of the evolutionary game that clonal populations play inside the body.
In the end, clonal populations turn the story of cancer from random bad luck into a predictable biological process that scientists are learning to interrupt. While the word “clonal” may sound intimidating on a report, it actually gives patients and doctors a clear map. One mutated cell starts the problem, but with modern tools we can watch the clones evolve, predict their next moves, and design treatments that keep them from taking over. This knowledge is already saving lives and will continue to improve outcomes as research moves forward. If you or a loved one has recently encountered the term clonal population in a cancer context, remember it is not just jargon; it is the key to understanding why the disease behaves the way it does and how we can outsmart it. For further clarification tailored to your specific pathology report, consider consulting experts at Honest Pathology, who specialize in making these complex concepts accessible and actionable for patients.
Image Credit: Manu Sharma