A brain tumor diagnosis often begins with one number that carries enormous weight: the WHO grade. That single digit from 1 to 4 tells doctors, and patients, how aggressively the tumor is likely to behave, how quickly it may grow, and what the realistic outlook might be. The World Health Organization’s grading system combines traditional microscopic features with modern molecular and genetic markers to create a more accurate picture than ever before. Lower grades usually mean slower growth and better long-term outcomes, while higher grades signal faster progression and more intensive treatment needs. Understanding your specific grade, including what it implies for prognosis, helps cut through the fear and focus on informed decisions about care. This guide explains the WHO brain tumor grading system in straightforward terms, with special emphasis on prognosis, the question patients and families most frequently ask after hearing their grade.
If any part of this explanation leaves you wanting more clarity, especially about your own pathology report, services like Honest Pathology offer virtual consultations with board-certified pathologists who review reports line by line, explain terms in plain language, and answer your specific questions directly.
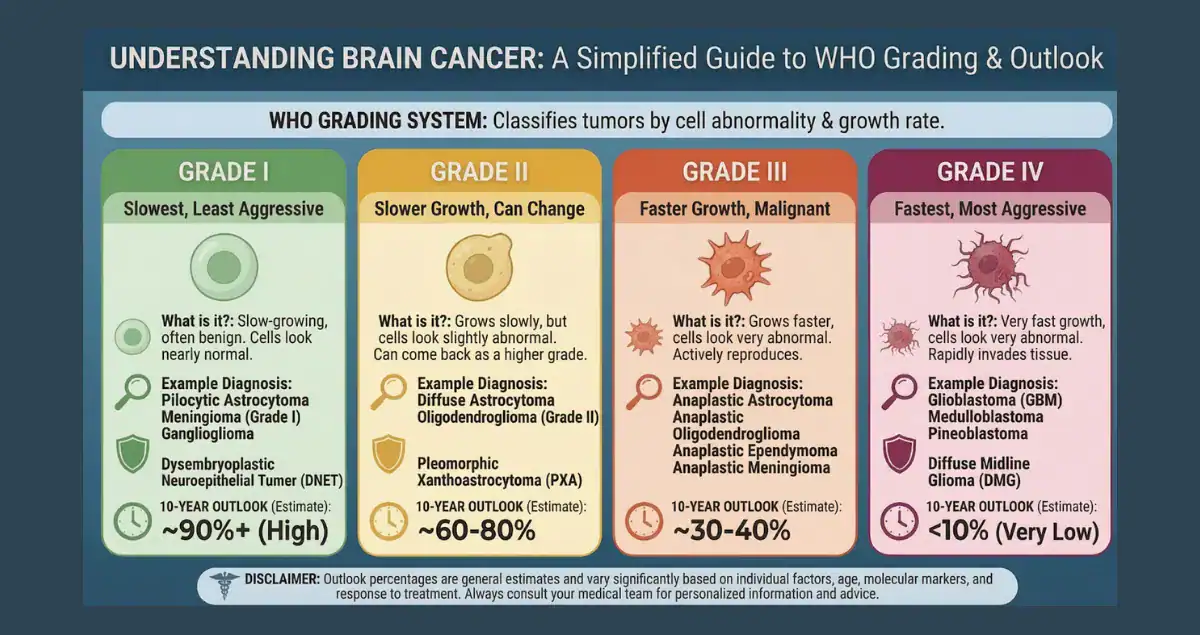
What Does WHO Grading Actually Mean for Brain Tumors?
The WHO grading system ranks brain tumors from 1 (least aggressive) to 4 (most aggressive) based on cell appearance, growth rate, and now genetic characteristics. Grade reflects not just how the tumor looks under the microscope but also its molecular profile, features like IDH mutation status, 1p/19q codeletion, or MGMT methylation that dramatically influence behavior and response to therapy. This integrated approach explains why two tumors with the same microscopic appearance can have very different prognoses. The grade is the single most powerful predictor of survival time, treatment intensity, and quality of life expectations.
How Do Doctors Determine the WHO Grade?
Grading begins with tissue obtained through biopsy or surgical resection. Pathologists evaluate four classic microscopic features: cell atypia (abnormality), mitotic activity (how often cells divide), microvascular proliferation (new blood vessel formation), and necrosis (areas of dead tissue). In the 2021 WHO classification, molecular testing is mandatory for many tumor types. For gliomas, absence of an IDH mutation can automatically upgrade a tumor to grade 4 even if it looks lower-grade microscopically. These combined findings produce the final grade, which is far more predictive of prognosis than older systems.
Grade 1 Brain Tumors: Excellent Prognosis with High Chance of Cure
Grade 1 tumors, such as pilocytic astrocytoma, subependymal giant cell astrocytoma, and many meningiomas, are typically slow-growing and well-circumscribed. Microscopically, the cells appear relatively normal with very low mitotic activity and no necrosis or vascular proliferation. Prognosis is excellent: complete surgical removal often results in cure, with 10-year survival rates exceeding 90–95% for most grade 1 tumors. Recurrence is rare after gross total resection, and many patients return to normal life with no long-term deficits. Even when complete removal is not possible due to location, these tumors often remain stable for decades with monitoring or targeted therapies.
Grade 2 Brain Tumors: Favorable but Guarded Long-Term Prognosis
Grade 2 tumors (diffuse astrocytoma, oligodendroglioma, ependymoma) grow slowly but infiltrate surrounding brain tissue, making complete removal challenging. Cells show moderate atypia and increased mitoses compared to grade 1, but no necrosis or prominent vascular changes. Prognosis varies widely depending on molecular features: IDH-mutant grade 2 gliomas carry a median survival of 7–15 years, while IDH-wildtype tumors behave more aggressively and may be reclassified closer to grade 3 or 4 behavior. With maximal safe surgery followed by watchful waiting, radiation, or chemotherapy, many patients enjoy long periods of stability, often 5–10 years or more before progression. The risk of eventual malignant transformation to higher grade remains a key concern.
Grade 3 Brain Tumors: More Aggressive with Moderate Prognosis
Grade 3 tumors, including anaplastic astrocytoma and anaplastic oligodendroglioma, display clear malignant features: high mitotic rate, significant atypia, and early microvascular proliferation. Prognosis is shorter than lower grades but still measured in years rather than months. IDH-mutant anaplastic astrocytomas have median survival of 3–7 years with standard treatment (surgery, radiation, and chemotherapy), while 1p/19q-codeleted anaplastic oligodendrogliomas often fare better, with medians exceeding 10 years in many series. IDH-wildtype grade 3 tumors carry poorer outcomes, sometimes overlapping with grade 4. Aggressive multimodal therapy can extend meaningful survival and maintain quality of life for several years.
Grade 4 Brain Tumors: Most Aggressive with the Shortest Prognosis
Grade 4 tumors, led by glioblastoma (IDH-wildtype), represent the highest level of malignancy. They show all aggressive histologic features plus frequent necrosis and robust vascular proliferation. Median survival for glioblastoma remains approximately 12–18 months with standard treatment (maximal resection, radiation, and temozolomide chemotherapy), though 5-year survival is around 5–10%. Favorable molecular markers, such as MGMT promoter methylation, can extend median survival to 20–30 months or longer in some cases. Younger patients, those with good performance status, and those eligible for clinical trials or tumor-treating fields (Optune) often achieve better-than-average outcomes. Other grade 4 tumors (e.g., grade 4 astrocytoma, IDH-mutant) may have slightly longer medians, but the overall prognosis remains guarded.
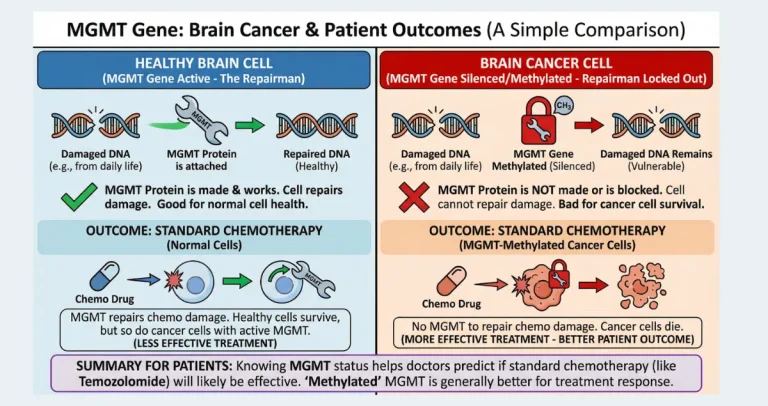
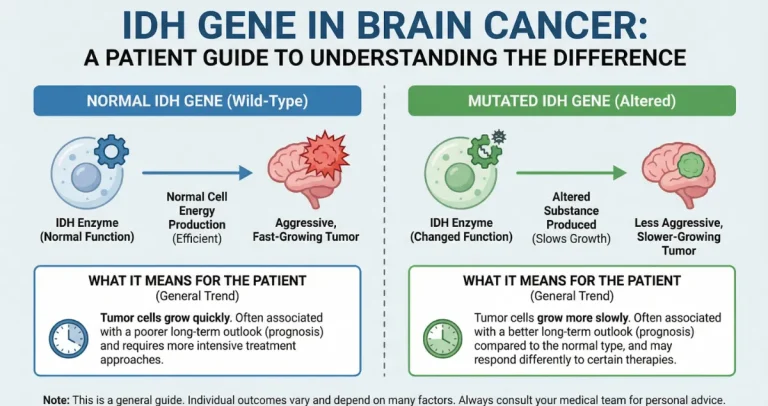
Why Molecular Features Now Dominate Prognosis Predictions
The 2021 WHO update shifted emphasis from pure histology to molecular-integrated grading. An IDH-mutant tumor that looks grade 2–3 microscopically has a far better prognosis than an IDH-wildtype tumor of similar appearance, which is now classified as glioblastoma (grade 4) regardless of histology. MGMT methylation predicts better response to temozolomide, while 1p/19q codeletion marks oligodendrogliomas with particularly favorable long-term survival. These markers explain why prognosis can vary dramatically within the same histologic grade and why genetic testing is now essential for accurate counseling.
How the Grade Influences Treatment Intensity and Prognosis
Grade directly dictates treatment aggressiveness. Grade 1 tumors often require only surgery and surveillance, leading to near-normal life expectancy. Grade 2 may involve observation, radiation, or chemotherapy to delay progression and preserve function. Grade 3 and 4 almost always demand multimodal therapy, surgery, radiation, chemotherapy, and increasingly targeted agents or immunotherapy trials. Higher grades correlate with shorter progression-free and overall survival, but modern therapies have meaningfully extended meaningful survival even in grade 4 cases. Regular MRI monitoring is standard across all grades to detect recurrence early, when additional treatment can still improve outcomes.
Can Prognosis Improve with Treatment or Change Over Time?
Prognosis is not fixed at diagnosis. Effective therapy can significantly prolong survival beyond the median, especially in lower-grade tumors or those with favorable genetics. Lower-grade tumors can undergo malignant progression to higher grade, worsening prognosis and requiring re-biopsy and intensified treatment. Conversely, molecularly favorable high-grade tumors sometimes respond exceptionally well, leading to longer-than-expected survival. Age, performance status, tumor location, extent of resection, and access to advanced therapies all modify the outlook.
The Diagnostic Timeline and When Prognosis Becomes Clear
From initial MRI to final integrated WHO grade usually takes 2–6 weeks. Basic histologic grade may be available within days of surgery, but full molecular profiling (IDH, 1p/19q, MGMT, etc.) adds 1–3 weeks. Doctors often discuss preliminary prognosis based on early findings while awaiting molecular confirmation, which can refine or even change the expected survival range.
In the end, the WHO grade is more than a number, it is the foundation for understanding prognosis, planning treatment, and setting realistic expectations. While higher grades carry shorter survival averages, individual outcomes vary widely thanks to molecular insights and advancing therapies. Knowing the full picture, including genetic markers, empowers patients and families to ask targeted questions and pursue the most appropriate care. If you or a loved one has a brain tumor pathology report and want detailed help interpreting the WHO grade, molecular results, and what they truly mean for prognosis and next steps, consider consulting experts at Honest Pathology, who specialize in translating complex reports into clear, personalized information for patients. This clarity helps you face the journey with greater confidence and control.