In the world of brain tumor pathology, few molecular findings carry as much practical weight as MGMT promoter methylation. When a pathology report mentions that the MGMT gene promoter is methylated (or “silenced”), it often signals better news than the diagnosis itself might suggest. MGMT stands for O6-methylguanine-DNA methyltransferase, a protein that repairs DNA damage caused by certain chemotherapy drugs like temozolomide (TMZ). When the promoter region of the MGMT gene is methylated, the gene is turned off, meaning less repair protein is produced. This makes tumor cells more vulnerable to TMZ, which adds methyl groups to DNA and triggers cell death if the damage cannot be fixed. This epigenetic change is one of the strongest predictors of how well patients with glioblastoma (GBM) and other high-grade gliomas respond to standard treatment. It also influences survival expectations and helps guide decisions about chemotherapy intensity. This guide explains MGMT promoter methylation in clear terms, addressing the questions most patients have after seeing the result: what causes it, how it is tested, why it predicts better outcomes, and what it means for real-world treatment and prognosis.
If any part of this explanation leaves you wanting more clarity, especially about your own pathology report, services like Honest Pathology offer virtual consultations with board-certified pathologists who review reports line by line, explain terms in plain language, and answer your specific questions directly.
What Is MGMT Promoter Methylation and Why Does It Happen in Brain Tumors?
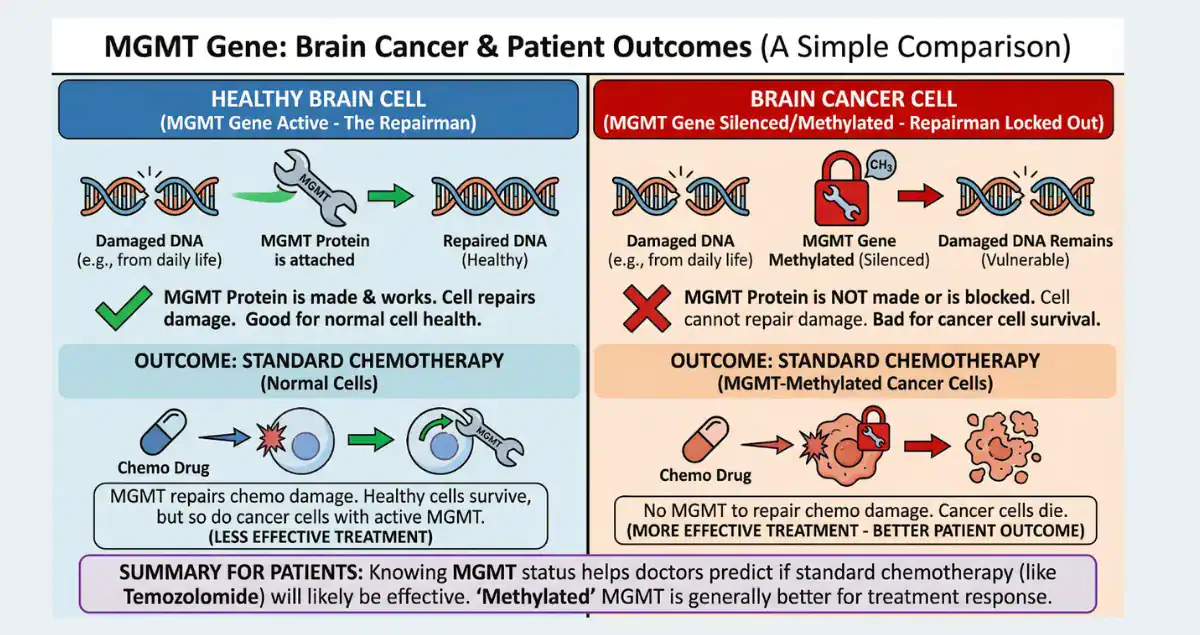
The MGMT gene helps cells fix specific types of DNA damage. In many gliomas, especially glioblastoma, the promoter region, a control switch upstream of the gene, becomes methylated, adding methyl groups that silence the gene. This epigenetic silencing reduces MGMT protein levels in tumor cells, impairing their ability to repair alkylation damage from drugs like TMZ. Methylation occurs early in tumor development and is found in about 40–50% of glioblastomas, higher rates (70–80%) in lower-grade and anaplastic gliomas, and nearly all IDH-mutant oligodendrogliomas. It is rare in non-glioma brain tumors or metastases. The change is not inherited but acquired during tumor formation, often linked to broader epigenetic dysregulation in cancer cells.
How MGMT Methylation Fits into the Modern WHO Classification and Diagnosis
The 2021 WHO classification integrates MGMT status as a key prognostic marker, though it does not directly alter tumor grade like IDH or 1p/19q changes do. In glioblastoma (IDH-wildtype, grade 4), MGMT methylation does not change the diagnosis but strongly predicts response to alkylating chemotherapy. Testing is routine for all newly diagnosed high-grade gliomas to guide therapy. It complements other markers: MGMT-methylated tumors often overlap with IDH-mutant gliomas (which already have better prognosis), but methylation provides independent benefit even in IDH-wildtype GBM.
Testing Methods for MGMT Promoter Methylation
MGMT status is determined from tumor tissue obtained during biopsy or resection. The most common methods include:
- Methylation-specific PCR (MSP): A sensitive, cost-effective technique that amplifies methylated vs. unmethylated DNA after bisulfite conversion. It is widely used but can be qualitative or semi-quantitative.
- Pyrosequencing (PSQ): Provides quantitative measurement of methylation at multiple CpG sites in the promoter, often considered more reliable for prognostication due to cutoff thresholds.
- Quantitative real-time MSP (e.g., MethyLight): Offers higher precision and is good for low DNA amounts from small biopsies.
- Other approaches like bead arrays, MS-MLPA, or next-generation sequencing are used in research or specialized labs.
Results are typically available within 1–3 weeks after surgery, depending on the lab. Immunohistochemistry for MGMT protein is less reliable due to variability and is not recommended as the primary method. A common question is about cutoffs: many labs use percentage methylation (e.g., >9–20% considered methylated), but quantitative methods like pyrosequencing help refine predictions.
Why MGMT Methylation Is a Powerful Prognostic and Predictive Biomarker
MGMT methylation is both prognostic (linked to better survival regardless of treatment) and predictive (stronger benefit from TMZ). Landmark trials showed that in GBM:
- Methylated tumors have median overall survival of 16–21 months with standard chemoradiation (surgery + radiotherapy + TMZ), vs. 11–12 months for unmethylated.
- Recent large cohorts (e.g., NCDB data) report median OS of 16.4 months for methylated vs. 11.8 months for unmethylated GBM, with 3-year survival around 24% vs. 10%.
- In elderly patients or those with poor performance status, methylated tumors may benefit from TMZ alone over radiotherapy alone.
The benefit stems from impaired DNA repair: TMZ adds damaging methyl groups that MGMT normally removes. Without sufficient MGMT, cells accumulate lethal damage and die. This effect is most pronounced in newly diagnosed GBM but also seen in recurrent disease and lower-grade gliomas treated with alkylators.
How MGMT Status Shapes Treatment Choices
Standard GBM treatment (Stupp protocol: maximal resection + radiotherapy + concurrent/adjuvant TMZ) is recommended regardless of MGMT status, as even unmethylated patients derive some benefit. However:
- Methylated tumors show greater gains from TMZ addition (median survival increase of 6–9 months in key studies).
- In elderly patients (>70 years), methylated status favors TMZ monotherapy over radiotherapy alone in some analyses.
- Unmethylated tumors may prompt earlier consideration of clinical trials, alternative regimens, or de-escalation to avoid toxicity if response is expected to be limited.
- Emerging targeted approaches (e.g., PARP inhibitors in combination) are under study for unmethylated cases to overcome resistance.
MGMT status also informs trial eligibility and personalized strategies, with ongoing research into demethylating agents to sensitize unmethylated tumors.
MGMT Methylation in Lower-Grade Gliomas and Other Contexts
In IDH-mutant grade 2–3 gliomas, MGMT methylation is common (especially in oligodendrogliomas) and associates with better response to TMZ or PCV chemotherapy. It adds prognostic value beyond IDH and 1p/19q status. In IDH-wildtype lower-grade tumors, methylation is less frequent but still predicts improved outcomes with alkylators.
The Diagnostic Timeline and Interpreting Your Report
MGMT testing follows surgery and is part of routine molecular profiling. Basic results (methylated vs. unmethylated) arrive in days to weeks; quantitative details may take longer. Reports often include percentage methylation or a categorical call. A common question is whether borderline results matter, quantitative methods help clarify gray zones. Even in unmethylated cases, standard treatment remains the backbone, with trials offering hope for future options.
In the end, MGMT promoter methylation is one of the most actionable findings in brain cancer pathology. When present, it turns a tough diagnosis into one where chemotherapy can meaningfully extend survival and improve quality of life. While glioblastoma remains challenging, this marker, identified over two decades ago—continues to guide better, more personalized care and fuels research into overcoming resistance. If your report includes MGMT results and you want clear breakdowns of what the methylation status truly means for your prognosis, TMZ benefit, or next steps, consider consulting experts at Honest Pathology, who specialize in translating molecular pathology details into understandable, empowering information for patients. This insight helps you discuss options confidently with your neuro-oncology team.