Brain cancer, particularly gliomas, remains one of the most challenging diagnoses a person can face, yet advances in molecular understanding have brought new hope to patients and families. Among the most significant breakthroughs is the discovery of IDH mutations in brain cancer, a genetic alteration that redefines how these tumors are classified, predicted, and managed. For those searching online about IDH mutations in brain cancer or what an IDH mutant glioma means for survival and options, the answers lie in how this single change alters tumor behavior at the deepest cellular level. Far from being just another lab result, IDH status guides everything from diagnosis to long-term care, often signaling a more favorable path compared to tumors without the mutation.
What Exactly Are IDH Mutations and Why Do They Matter in Brain Tumors?
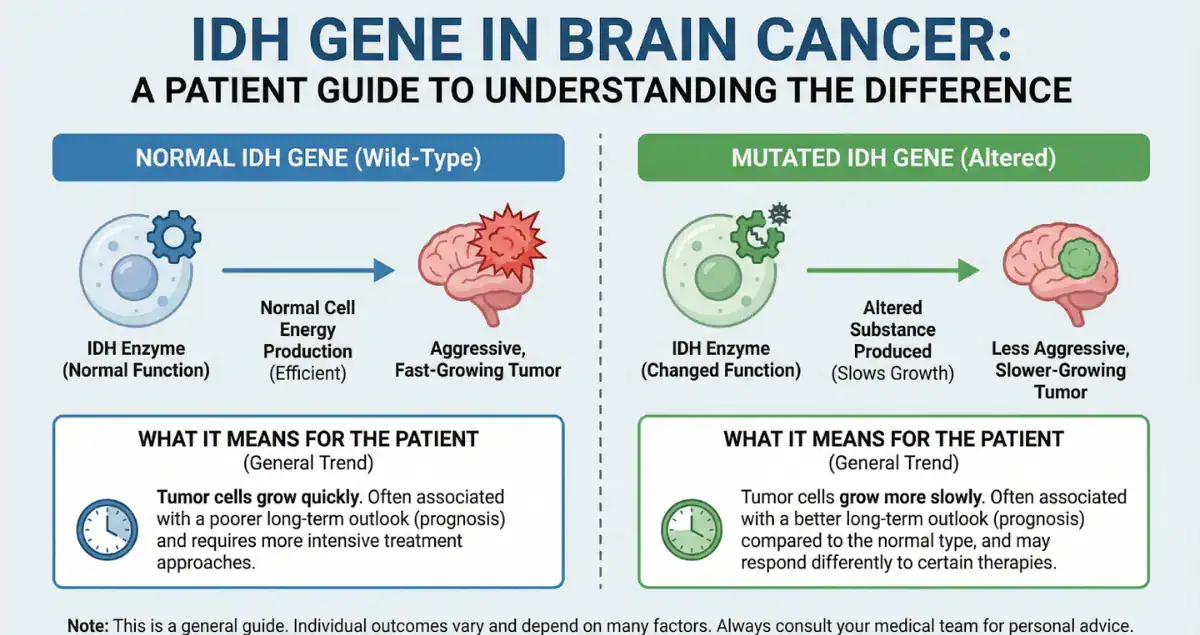
Isocitrate dehydrogenase, or IDH, refers to enzymes that play a routine role in cellular metabolism, helping convert nutrients into energy through the Krebs cycle. When a mutation occurs in the IDH1 or IDH2 gene, typically a somatic change acquired in the tumor cells rather than inherited, the enzyme gains a new, harmful function. Instead of producing normal energy metabolites, the mutated version churns out an abnormal byproduct called 2-hydroxyglutarate, or 2-HG. This oncometabolite disrupts normal DNA and histone regulation, leading to widespread epigenetic changes that trap cells in a primitive, proliferative state and promote tumor formation. In the context of brain cancer, these mutations appear early in the disease process for many gliomas, making them a foundational driver rather than a late complication. Patients frequently ask whether IDH mutations are common or rare in brain tumors; the reality is that they occur in roughly 70 to 80 percent of lower-grade gliomas and a smaller subset of higher-grade cases, reshaping the entire landscape of adult-type diffuse gliomas.
How IDH Mutant Gliomas Differ from IDH Wildtype Brain Cancers
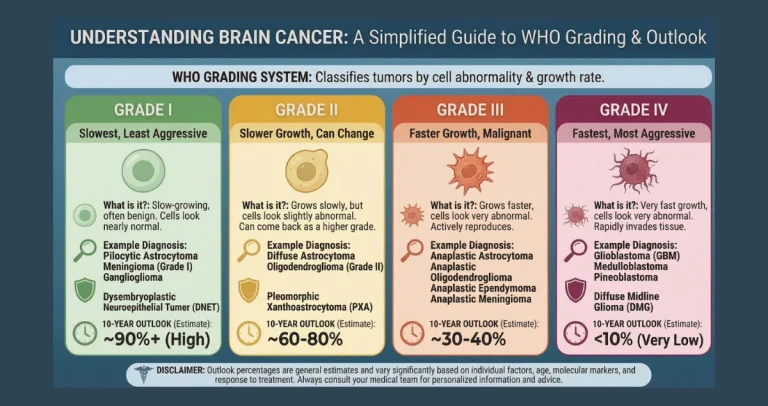
The distinction between IDH mutant and IDH wildtype gliomas is now central to modern classification under the World Health Organization guidelines. Tumors harboring the mutation tend to arise in younger adults, grow more slowly, and respond differently to therapies than their wildtype counterparts, which are often aggressive primary glioblastomas. IDH wildtype tumors, by contrast, lack this alteration and frequently behave like high-grade malignancies from the outset, with rapid infiltration and poorer responses to standard treatments. This molecular split explains why two gliomas that look similar under a microscope can have vastly different clinical courses. Many individuals researching IDH mutation brain cancer prognosis discover that the presence of the mutation consistently correlates with extended survival across grades, turning what was once a uniformly grim outlook into one with meaningful years ahead. For instance, secondary glioblastomas that evolve from lower-grade tumors and carry the mutation are now reclassified separately from primary IDH wildtype glioblastoma, highlighting the prognostic power of this marker.
The Critical Role of Accurate IDH Testing in Diagnosing Brain Cancer
Detecting IDH mutations begins with tissue obtained during surgery, where neuropathologists use a combination of techniques to determine status. The most common alteration, IDH1 R132H, can be spotted quickly through immunohistochemistry, a specialized staining method that highlights the mutant protein. For rarer variants, next-generation sequencing provides a comprehensive view, ensuring no subtle changes are missed. This testing is not optional; it directly informs the tumor’s official name and grade, such as astrocytoma IDH-mutant or oligodendroglioma defined by both the mutation and 1p19q chromosomal codeletion. Without precise molecular profiling, patients risk misclassification that could affect treatment choices and expectations. Those wondering how doctors confirm IDH status in brain cancer or whether a second opinion on pathology matters quickly learn that accuracy here is everything. Many turn to specialized services like Honest Pathology for their transparent, expert-level molecular analysis that delivers clear, reliable results tailored to guide the next steps with confidence.
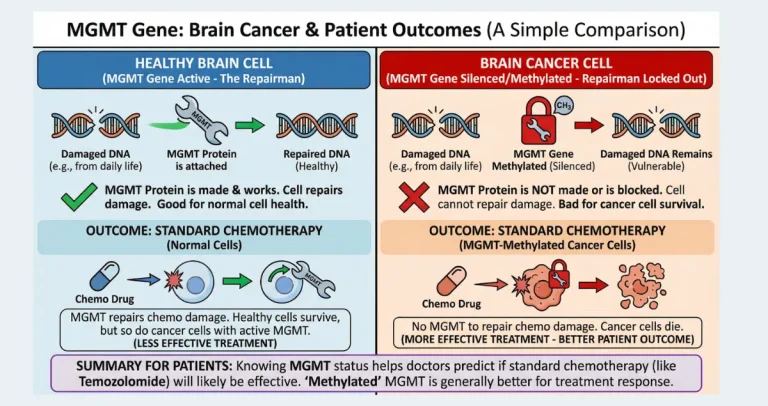
How IDH Mutations Shape Prognosis and Long-Term Survival in Gliomas
One of the most reassuring aspects for patients diagnosed with an IDH mutant glioma is the consistently better outlook compared to wildtype disease. These tumors often progress more slowly, allowing for extended periods of stability even after initial surgery. Median survival figures tell a compelling story: individuals with IDH-mutant grade 3 astrocytomas, for example, may live five years or longer in many cases, versus significantly shorter intervals for similar-grade wildtype tumors. Even in higher-grade settings, the mutation confers advantages in response to therapy and overall disease control. Factors such as age, extent of resection, and additional markers like 1p19q codeletion or MGMT promoter methylation further refine predictions, but IDH status remains the strongest independent indicator of prolonged survival. Families researching IDH mutation brain cancer survival rates often feel a sense of relief upon learning this detail, as it shifts conversations from immediate crisis to strategic, long-term management. The slower growth pattern also means symptoms like seizures or headaches may appear earlier and be more manageable, giving patients valuable time to adapt and pursue active lives.
Standard Treatment Strategies for IDH-Mutant Brain Tumors
Management of IDH-mutant gliomas typically starts with maximal safe surgical resection, aiming to remove as much tumor as possible while preserving neurological function. Following surgery, decisions about radiation and chemotherapy depend on grade, residual disease, and individual risk profile. Low-risk grade 2 tumors might be monitored with regular imaging in select cases, while higher-risk or grade 3 lesions often proceed to combined radiation and chemotherapy regimens such as PCV or temozolomide. These approaches exploit the heightened sensitivity many IDH-mutant tumors show to alkylating agents, leading to durable responses. Patients frequently seek clarity on whether IDH status changes everyday treatment protocols, and the answer is yes in subtle but important ways: the mutation helps oncologists tailor intensity and timing to balance efficacy with quality of life. Ongoing surveillance through MRI scans becomes a routine part of the journey, allowing early detection of any changes without unnecessary intervention.
Breakthrough Targeted Therapies Targeting IDH Mutations in Brain Cancer
The most exciting development in recent years has been the emergence of therapies designed specifically to inhibit the mutant IDH enzyme and block 2-HG production. Vorasidenib, a dual IDH1 and IDH2 inhibitor, stands out as a landmark option approved for grade 2 astrocytomas and oligodendrogliomas following surgery. Clinical data show it dramatically extends progression-free survival, often doubling the time before further treatment is needed while maintaining a favorable safety profile. Other inhibitors, including ivosidenib, continue to be explored in trials for recurrent or higher-grade disease, offering hope that targeted approaches can delay or reduce reliance on traditional radiation and chemotherapy. For those asking about new treatments for IDH mutation brain cancer or whether targeted drugs are available, these advancements represent a shift from generic strategies to precision medicine that directly addresses the tumor’s molecular driver. Real-world experience continues to affirm their role in preserving function and extending stable periods, particularly for patients with residual low-grade disease.
Navigating Life with an IDH-Mutant Glioma: Practical Insights for Patients
Living with an IDH-mutant brain tumor involves more than medical appointments; it means embracing a future that, while requiring vigilance, often includes meaningful milestones. Seizure management, cognitive support, and lifestyle adjustments become key, yet many patients report sustained quality of life thanks to the slower disease pace. Participation in clinical trials exploring combinations of IDH inhibitors with immunotherapy or other agents opens additional doors, and support networks help families stay informed. Common concerns about inheritance or family risk are usually put to rest, as these mutations are overwhelmingly tumor-specific rather than germline. Regular molecular re-testing at progression ensures treatments evolve with the disease. Throughout this process, having an accurate foundation in pathology remains essential, which is why many individuals and their care teams choose Honest Pathology for thorough, honest interpretations that empower confident decision-making.
Looking Ahead: Hope and Progress in IDH-Mutant Brain Cancer Care
The story of IDH mutations in brain cancer illustrates how one molecular insight can transform an entire field. What once seemed like an inscrutable disease now yields to targeted strategies that extend survival and improve daily living. As research continues to refine classifications, uncover new combinations, and expand access to inhibitors, patients today stand on firmer ground than ever before. Whether newly diagnosed or years into the journey, understanding IDH status provides both practical guidance and genuine optimism. For anyone seeking clarity on their brain tumor pathology or the latest implications of IDH mutations, partnering with trusted experts ensures no detail is overlooked. Honest Pathology offers precisely that level of specialized support, delivering the reliable molecular insights that help turn complex diagnoses into personalized roadmaps forward.